Curious to know how an absorption chiller works? Don’t worry we’ve got you covered.
For beginners, an absorption chiller is a closed loop cycle that uses waste heat to provide cooling or refrigeration.
This absorption cooling cycle relies on three basic principles:
- When a liquid is heated it boils (vaporizes) and when a gas is cooled it condenses
- Lowering the pressure above a liquid reduces its boiling point
- Heat flows from warmer to cooler surfaces.
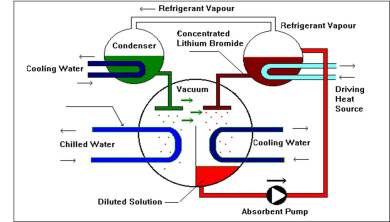
The process of absorption cooling is dependent on a thermochemical ‘compressor’. Two different fluids are used: a refrigerant and an absorbent. The fluids have high “affinity” for each other, which means one dissolves easily in the other.In a water-lithium bromide vapour absorption refrigeration system, water is used as the refrigerant while lithium bromide (Li Br) is used as the absorbent. In the absorber, the lithium bromide absorbs the water refrigerant, creating a solution of water and lithium bromide.
The refrigerant—usually water—can change phase easily between liquid and vapour and circulates through the system.
Heat from natural gas combustion or a waste-heat source drives the process. The high affinity of the refrigerant for the absorbent (usually lithium bromide or ammonia) causes the refrigerant to boil at a lower temperature and pressure than it normally would and transfers heat from one place to another.
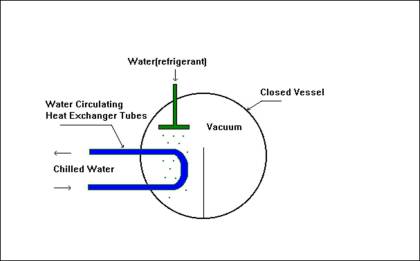
STEP 1
Boiling point of the water acts as a function of pressure. At atmospheric pressure, water boils at 100 deg. C. When maintained at high vacuum, water will boil and sub-cool itself. The boiling point of the water at 6 mm-Hg (abs) is 3.7 deg.C.
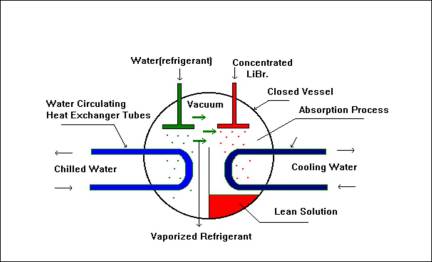
STEP 2
Lithium Bromide (LiBr) has the property to absorb water (Refrigerant) due to its chemical affinity. It is directly proportional to concentration and inversely proportional to its temperature.
STEP 3
Diluted LiBr loses its capacity to absorb water vapour. Thus, needs to be re-concentrated using a Heat Source. Heating may be in the form of Steam / Flue gases / Hot water /fuel firing.
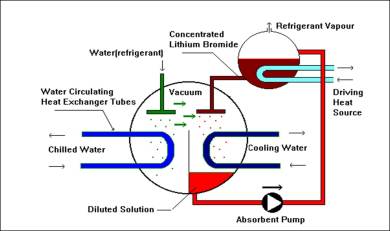
STEP 4
The heating causes the solution to release the absorbed refrigerant in the form of vapour. This vapour is cooled in a separate chamber to become liquid Refrigerant.

Please if some one can answer me , what is the role of cooling water in the absorber
LikeLike
In the absorber, the cooling water removes the heat of condensation and dilution before rejecting it to a cooling tower.
LikeLike
thanks
LikeLike
I wanted to thank you for explaining absorption chillers. I actually didn’t know the absorbent could be lithium bromide or ammonia. I’m kind of interested to learn ho much of the absorbent is used or if that depends on the size of the chiller.
LikeLike